Caffeine structure with lone pairs10/2/2023 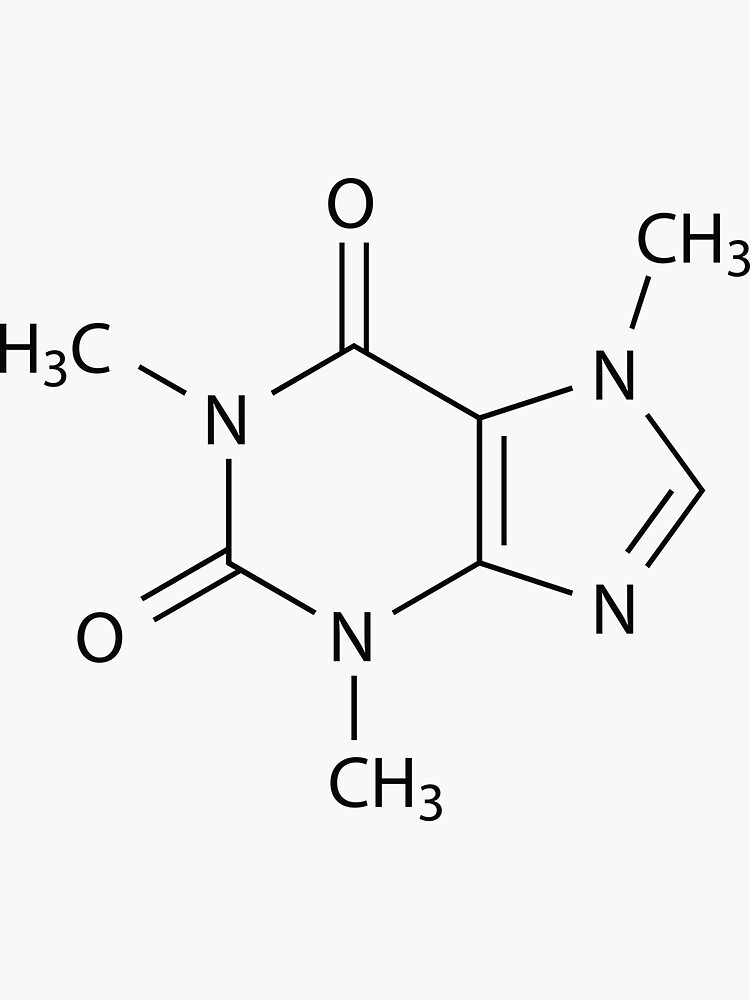 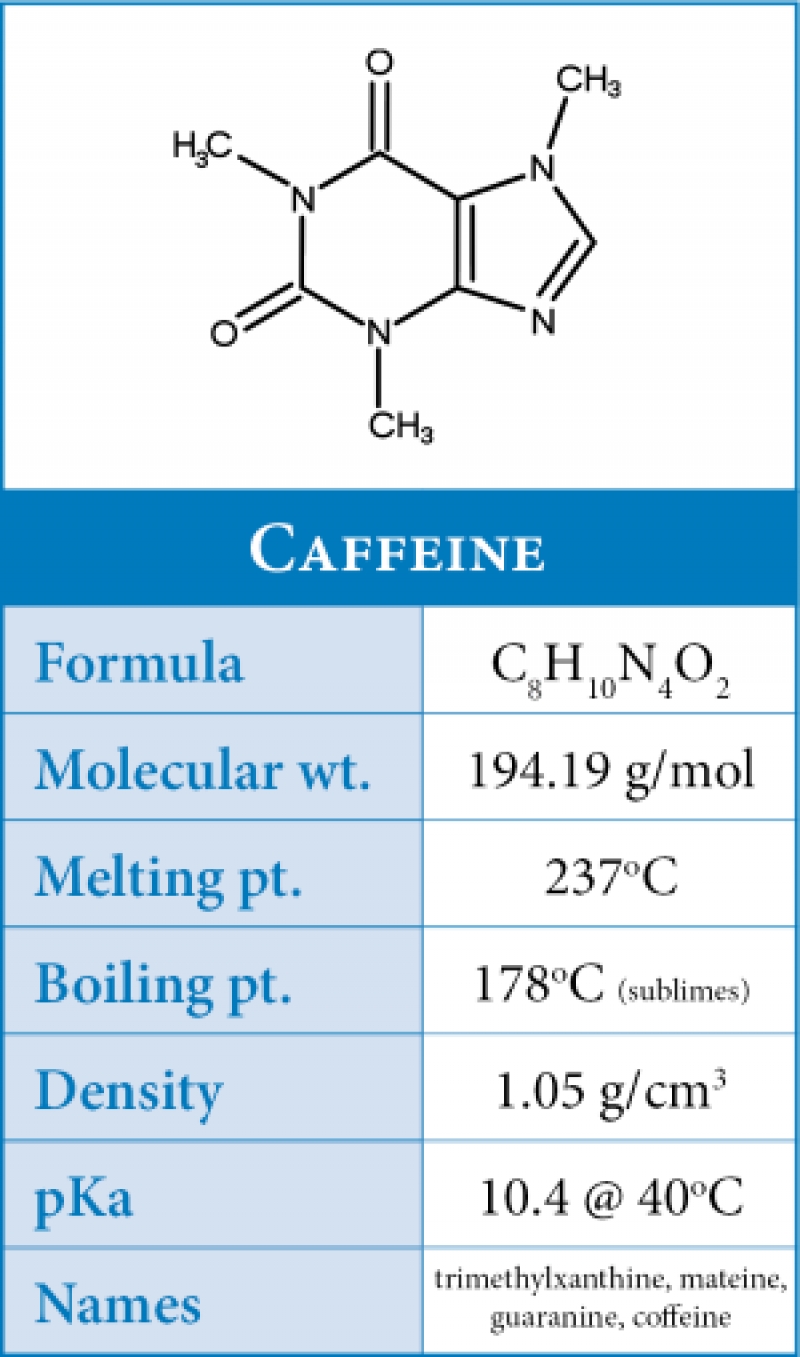 
The Lewis structure has three bonded pairs and two lone pairs for a total of 5 groups of charge so we predict a trigonal bipyramidal structure with the lone pairs which occupy more space on the equatorial positions. The two F's are equivalent so one NMR peak is expected. The Lewis structure has 2 bonded pairs and two lone pairs for a total of 4 groups of charge so a tetrahedral structure is expected with a FIF bond angle close to 109.5 degrees. Lewis dot structures and sketches of structures Predict in each case the number of peaks in the 19F NMR spectrum. Draw the Lewis structure and predict the three-dimensional structure of the following molecular species.The species is an anion and charge must reside on one of the atoms. Recall oxygen is more electronegative than bromine. The best structures place zero formal charge on the Br atom and a formal charge of Give the basis for your selection in the second part.The remaining 4 structures are valid but the 2nd and third structures, the resonance structures, are the best. Draw a circle around the best structure(s).The two structures at the right have more than 8 electrons around an oxygen atom and violate the Pauli Exclusion Principle. Draw a large "X" through any invalid structure(s).Lewis electron-dot structures of the bromite anion, BrO 2 -, are drawn below.The high melting point of uranium trifluoride indicates ionic bonding and the strong electrostatic forces between the ions in the lattice. Ionic bonding is energetically very unlikely as the cost of producing a U +6 cation is too high. high vapor pressure, of uranium hexafluoride indicates covalent bonding, strong intramolecular forces but weak intermolecular forces. Account for the very striking differences of these fluorine compounds. The remarkably high volatility of uranium hexafluoride is the basis for the separation of the isotopes of uranium by the method of gaseous diffusion whereas uranium trifluoride has a very high melting point (>1000✬).However, with 3rd and high row elements, extended valence is possible and more than 8 electrons are permitted around an atom so quadruple bonds are possible and in fact have been observed. This violates the Pauli exclusiuon Principle. In most cases a quadruple bond will result with more than 8 electrons around an atom, e.g. Excluding the diatomic molecules X 2 (X = C, Si, Ge, Sn, and Pb) which require special treatment, are quadruple bond possible in stable compounds of 2nd row elements? of other elements? Explain.The overlap of the 2p z orbitals forces global planairty. The 2p z orbitals on these atoms which are adjacent to one another can form MO's and a system of delocalized electrons. Provide an explanation for this result.Īll heavy atoms in the ring and the 2 oxygens can or are sp 2 hybridized. All the heavy (non-hydrogen) atoms in caffeine lie in the same plane. Give the hybridization of the following atoms:.C 7N 8C 9, three groups of charge around N 8, hence, ca.O 4C 5C 6, 3 groups of charge around C 5, hence, ca.H 1C 2H 3, 4 groups of charge aound C 2, hence, ca.Estimate the bond angles defined by the following sets of atoms:.Is the one which places zero formal charge on the atoms. The structure given in the answer is only one of many resonance structures but it Correct the structure by drawing in the missing bonds. Some of the bonds may be double or triple bonds.In the molecular structure of caffeine, C 8H 10N 4O 2, which was given on the exam, all bonds were shown as single bonds. Solutions to the Second Midterm Examination
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
